Our research group focuses on the theoretical and computational investigation of materials and processes related to next-generation energy storage and conversion technologies. Using atomistic modeling, we seek to establish fundamental structure–property relationships that govern ionic transport, electrochemical stability, and mechanical robustness in battery materials, including electrolytes and electrode materials. A central theme of our work is the rational design of advanced electrode materials and soft solid electrolytes with enhanced ionic conductivity and long-term stability.
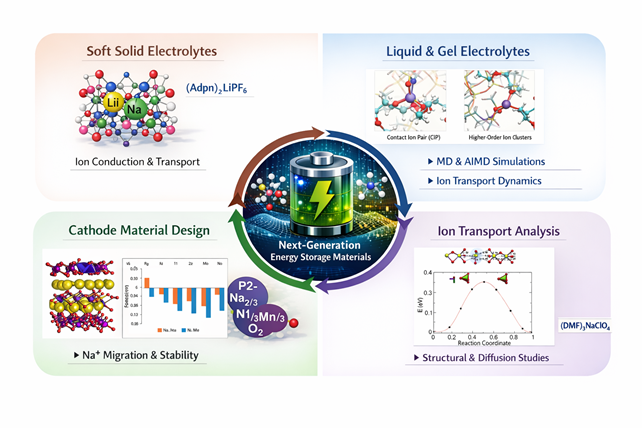
Soft Solid Electrolytes
In collaboration with experimental groups led by Prof. Michael Zdilla and Prof. Stephanie Wunder (Temple University, Philadelphia, USA), we study the structural and transport properties of soft solid co-crystalline battery electrolytes. Our work focuses on understanding ion coordination, lattice dynamics, and transport mechanisms in both lithium- and sodium-based soft electrolytes. Representative systems include lithium electrolytes such as (Adpn)2LiPF6 and (Adpn)2LiSbF6, as well as sodium electrolytes like (DMF)3NaClO4.
Liquid and Gel Electrolytes
We investigate the microscopic structure and ion-transport dynamics in lithium- and sodium-ion battery electrolytes through detailed analysis of ion–ion and ion–solvent interactions by Classical molecular dynamics (MD) and ab initio MD. By combining MD simulation with transport models, we quantify diffusion mechanisms, solvation structures, and ionic conductivity of NaPF6, POSS, LiFSI in carbonate and ether-based solvents.
Computational Design of Cathode Materials
We investigate the theoretical design of P2-type Na₂/₃Ni₁/₃Mn₂/₃O₂ (NNMO) cathode materials for sodium-ion batteries using computational methods. P2-NNMO is a promising layered transition-metal oxide cathode for SIBs. Using first-principles calculations and classical atomistic simulations.
We investigate the influence doping on crystal chemistry, cation ordering, and compositional tuning on Na⁺ migration, phase stability, and electrochemical performance.